 |
| Mouse Memories (source) |
Garner et al., (2012) use the same kind of genetic trickery that Han et al. (2009) used to erase memories. They genetically modified mice to express a foreign receptor that mice don't normally express. These kinds of receptors are called DREADDs which stands for "Designer Receptor Exclusively Activated by a Designer Drug." A DREADD can activate the cell, inactivate the cell, or even kill the cell. (Han et al., added a receptor that killed the the cells, but Garner et al. add a receptor than activates the cells.)
But, here's the real genetic trickery, the DREADD is promoted only in the cells that are active at a certain time. When something happens, the cells that are active during the event will express the DREADD. So later, when the designer drug is applied, only the cells which were active during the event will respond.
Using this DREADD system, Garner et al. try to trick mice into thinking that they were shocked in one room, when really they were shocked in another room. They call this a 'generating a synthetic memory trace' and this is how they do it:
 |
| Garner et al., 2012 Figure 2A |
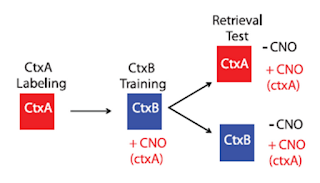
They have two rooms, context A (Ctx A) and context B (Ctx B). First they take the mouse and put it in context A (but don't give it an electric shock). This activates a certain subset of neurons and so the DREADD will get expressed in those neurons. Let's call them the "Context A neurons." Then they stop the creation of new DREADDs by adding in doxycyclin, which turns off the DREADD gene expression. This makes it so (in theory) the only cells that have DREADDs are the "Context A neurons."
Then they put the mouse in context B, but at the same time they apply the "designer drug" to activate the DREADDs. Since the DREADDs are (supposedly) only in the "Context A neurons," the neurons that the drug activates should trick the mouse into thinking it is actually in context A, when really it is in context B. Then they apply the shock to the mouse.
To see if they have 'generated a synthetic memory trace' the authors test whether the mouse freezes in context A (where it thinks it was shocked) or context B (where it was actually shocked).
 |
| Garner et al., 2012 Figure 2B&C |
Unfortunately the authors don't find something simple. First of all, they find that the mice with the DREADDs (the filled black circles above) almost always freeze less than the normal control mice (grey triangles), and they don't really explain why that might be. Second of all, they find that the application of the designer drug (+CNO) increases freezing for the DREADD mice in both context A and context B.
The mouse didn't learn that Context A is where it got shocked. Instead it learned that Context B with the "Context A neurons" is where it got shocked. It's like the "Context A neurons" become part of context B.
The authors call this a 'hybrid memory trace' where the mouse learns to associate a combination of the "Context A neurons" and the actual context B environment with the shock.
So what if just adding this drug is enough to create a hybrid memory? The authors did a nice control experiment to test this. They did the exact same protocol, but put the mouse in context B every single time (never in context A). That way the neurons expressing the DREADD are the "Context B neurons" and should basically be the same set of neurons that are active anyway when the mouse is shocked in Context B. In this case, the mouse froze a lot to context B without the drug, and it froze the same amount to context B with the drug. The drug caused no enhancement when it was activating the "Context B neurons." This is strong evidence that the hybrid memory trace has to involve the activation of a new set of neurons.
This is a really nice experimental design, but I think that the authors oversold their result a little bit in the title "Generation of a Synthetic Memory Trace." They didn't create a totally fake memory, they created a hybrid memory by adding in new neurons to the 'context' that the animal associated with the shock. There is no evidence that the mouse thought it was in context A or even that having a context A is important. If they had just stimulated a random, but new, set of neurons in context B and then stimulated that same random set of neurons when testing the mouse for freezing behavior, they might have seen the same results.
© TheCellularScale









